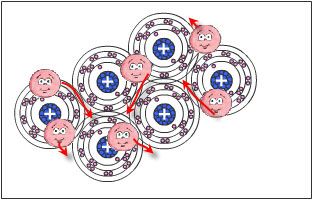
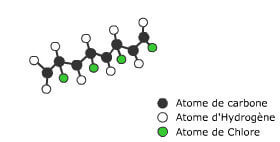
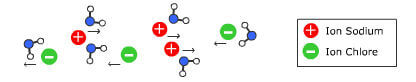
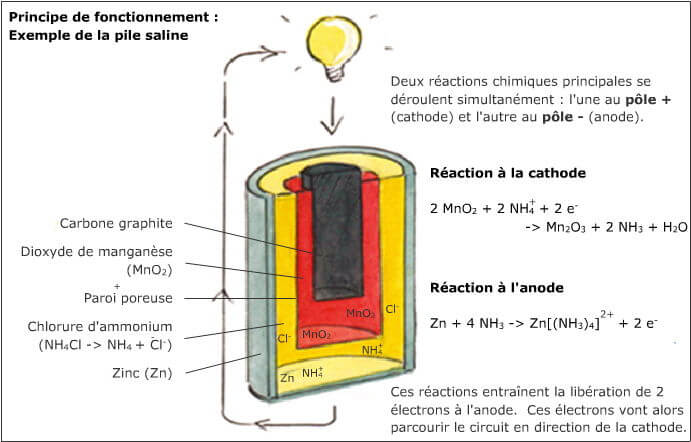
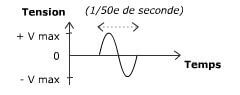
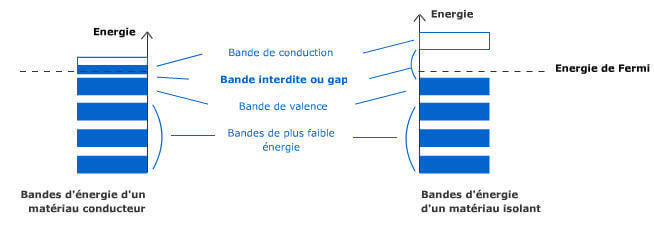
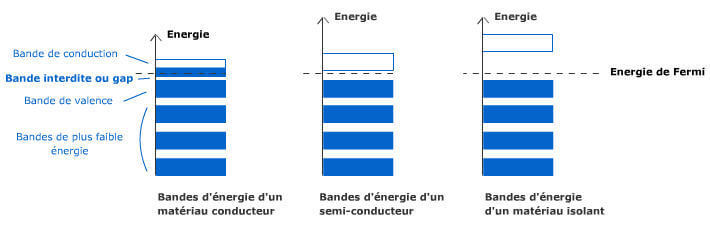
We use everyday numerous forms of energy: petroleum, wind, water, the sun are all energy vectors, just like our own body energy and that of animals. This energy gives us the means to produce work (e.g. movement, light or heat).
Among these different forms of energy, there is one which uses the energy of electrons: it is called Electricity. Its generation, its transmission and its use are possible because of its electromagnetic characteristics. Electricity and electromagnetism are tightly linked. In this module, we intend to have you (re)discover the basic concepts in electricity that you need to have in mind in order to tackle the subject of electromagnetism and electric and magnetic fields without apprehension.
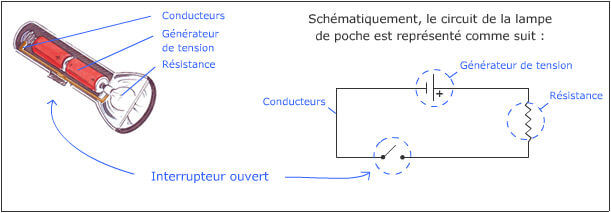
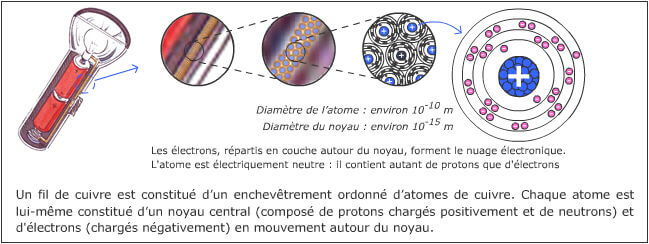
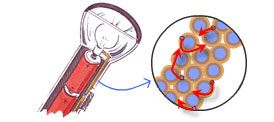
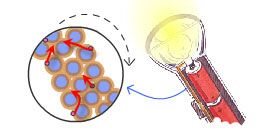
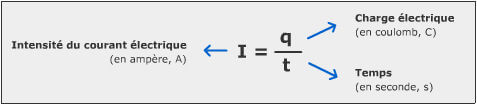
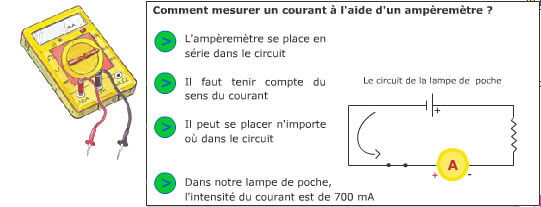
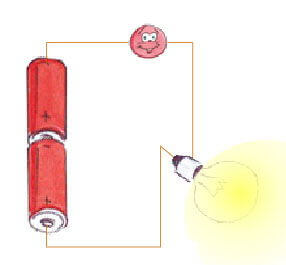
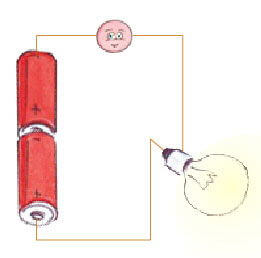
To get started, here is an illustration of a very simple electrical circuit: that of a flashlight.